Manufacture of Ophthalmic and Contact Lens Solution
Contact lens solutions include different products which perform a range of functions including cleaning, disinfecting, lubrication and rinsing. Ophthalmic solutions include safety eyewash and prescription preparations as well as “over the counter” medicinal products.
These solutions tend to have a similar base formulation: a saline solution (typically 5%) with a range of added ingredients according to the desired function. These include thickening agents, surfactants and active ingredients.

The Process
The process equipment must be capable of meeting the following process requirements:
- The mixer must provide vigorous in-tank movement to incorporate light powders which tend to float and to prevent salt granules from sinking to the bottom of the vessel.
- Some ingredients, (e.g. thickening agents and active ingredients) only make up a very small proportion of the formulation, but must be correctly dispersed and/or hydrated to obtain functionality.
- Many thickening agents are affected by salts, making it difficult to obtain functionality. This may be overcome by hydrating these ingredients before the salts are added, or by preparing them separately as a premix.
- The finished product is sterilized, typically by ultrafiltration. The solids must be fully solubilized and/or hydrated to ensure they are not removed during this process.
The Problem
Preparing saline solutions is a fairly simple task which can be adequately carried out using a conventional agitator. However, several problems can be encountered:
- The process must be carried out in the most sanitary manner possible. Problems can arise with conventional systems as they tend to allow a build-up of partially hydrated powder on vessel walls and mixer shafts etc. raising potential cleanliness issues.
- Stabilizers tend to form agglomerates which cannot be broken down by agitation.
- Long mixing times are required to fully hydrate and dissolve the solids. This can cause an undesired temperature rise, increasing the risk of bacterial contamination.
The Solution
These problems can be overcome and process times can be dramatically cut by using a Silverson High Shear mixer. Operation is as follows:

Stage 1
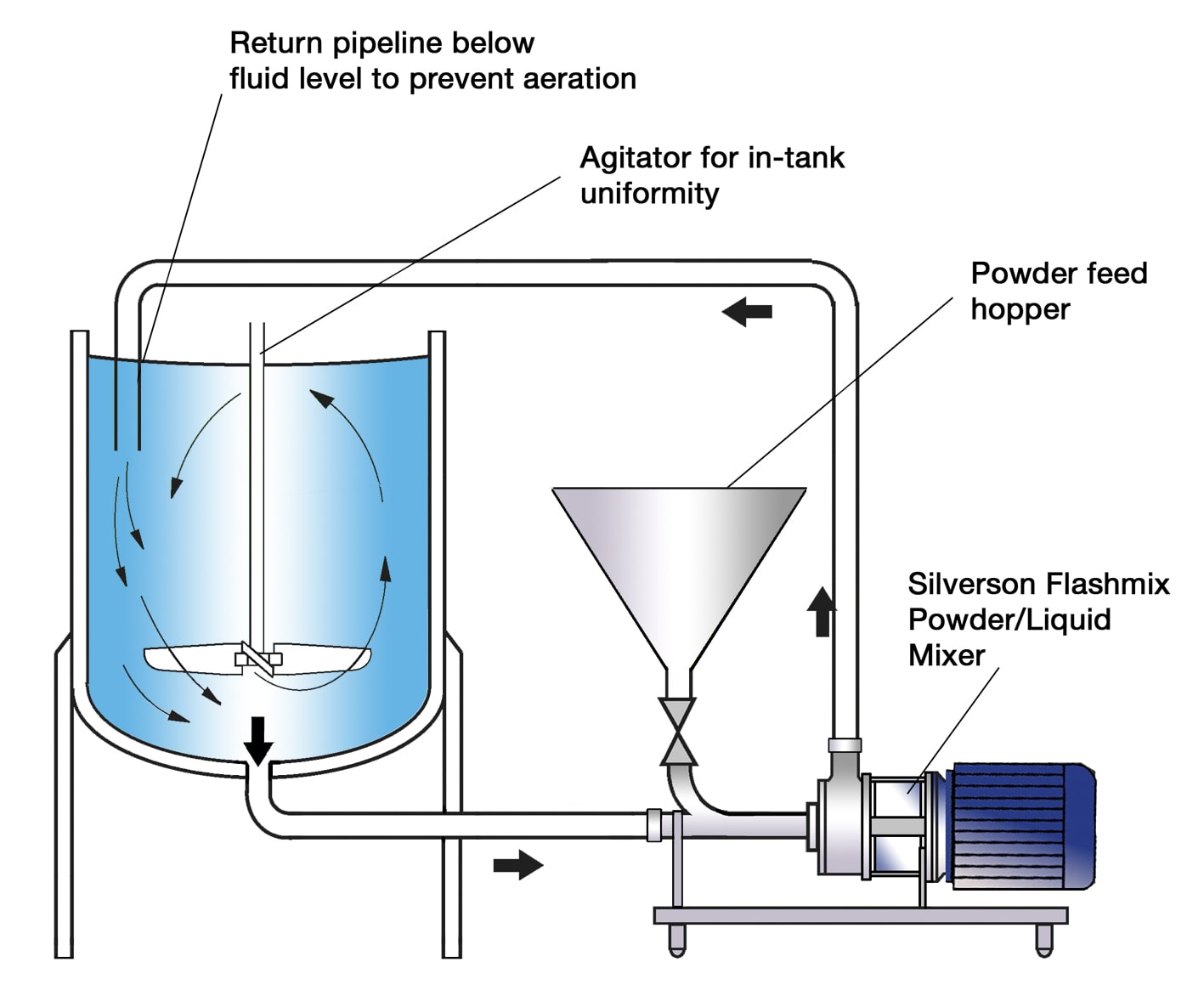
The vessel is charged with water. The mixer is started and the powders are added. The high speed rotation of the rotor creates a powerful suction which draws the liquid and solids upwards from the base of the vessel into the workhead.

Stage 2
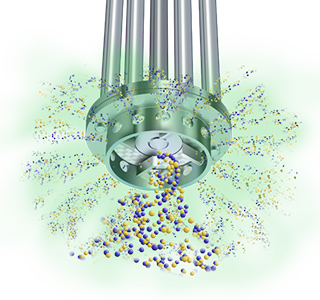
Centrifugal force then drives the materials to the periphery of the workhead where they are subjected to a milling action in the gap between the rotor and stator, rapidly reducing granule size. The product is then forced out through the stator at great velocity as fresh materials is drawn in.
Stage 3
Each pass through the workhead progressively reduces granule size and exposes an increasing surface area to the water, accelerating the solubilization of the salts, and ensuring that viscosity modifiers and active ingredients are fully dispersed and/or hydrated.
-
Stage 1

Stage 1
The vessel is charged with water. The mixer is started and the powders are added. The high speed rotation of the rotor creates a powerful suction which draws the liquid and solids upwards from the base of the vessel into the workhead.
-
Stage 2

Stage 2
Centrifugal force then drives the materials to the periphery of the workhead where they are subjected to a milling action in the gap between the rotor and stator, rapidly reducing granule size. The product is then forced out through the stator at great velocity as fresh materials is drawn in.
-
Stage 3

Stage 3
Each pass through the workhead progressively reduces granule size and exposes an increasing surface area to the water, accelerating the solubilization of the salts, and ensuring that viscosity modifiers and active ingredients are fully dispersed and/or hydrated.
The Advantages
This offers a number of advantages:
- Reduced CIP requirements.
- Processing time is rapidly reduced.
- Rapid incorporation and wetting out of powders.
- Greatly improved product consistency and stability.
- Easy validation.
The batch size, formulation and type of ingredients dictate which machine from the Silverson product line is most suitable:
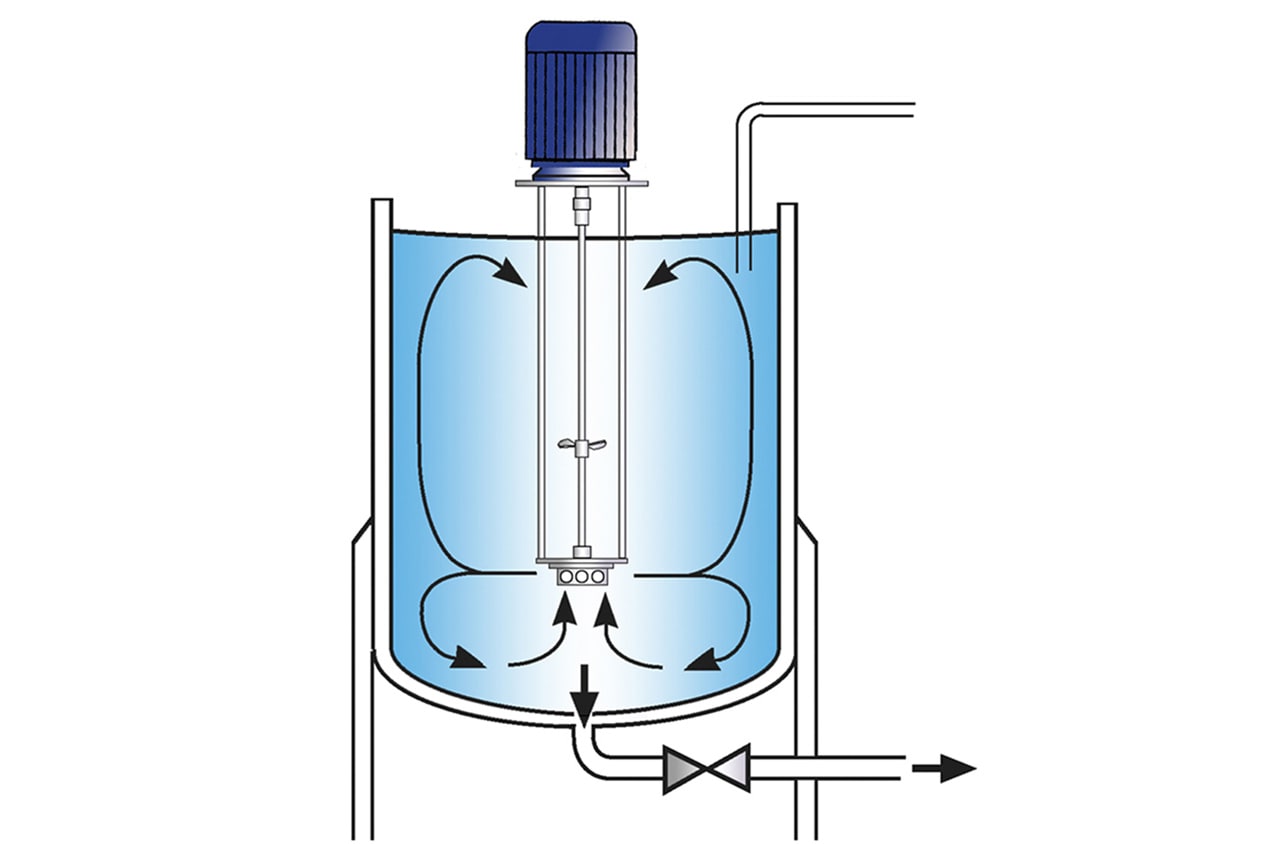
High Shear Batch Mixers
- Suitable for batch sizes up to 400 US gallons
- Can be vessel mounted or used on mobile floor stands
- Sealed units available for pressure/vacuum operation
- Small units available for R&D and pilot production
- Ultra Sanitary models available
- Can be supplied with FDA documentation
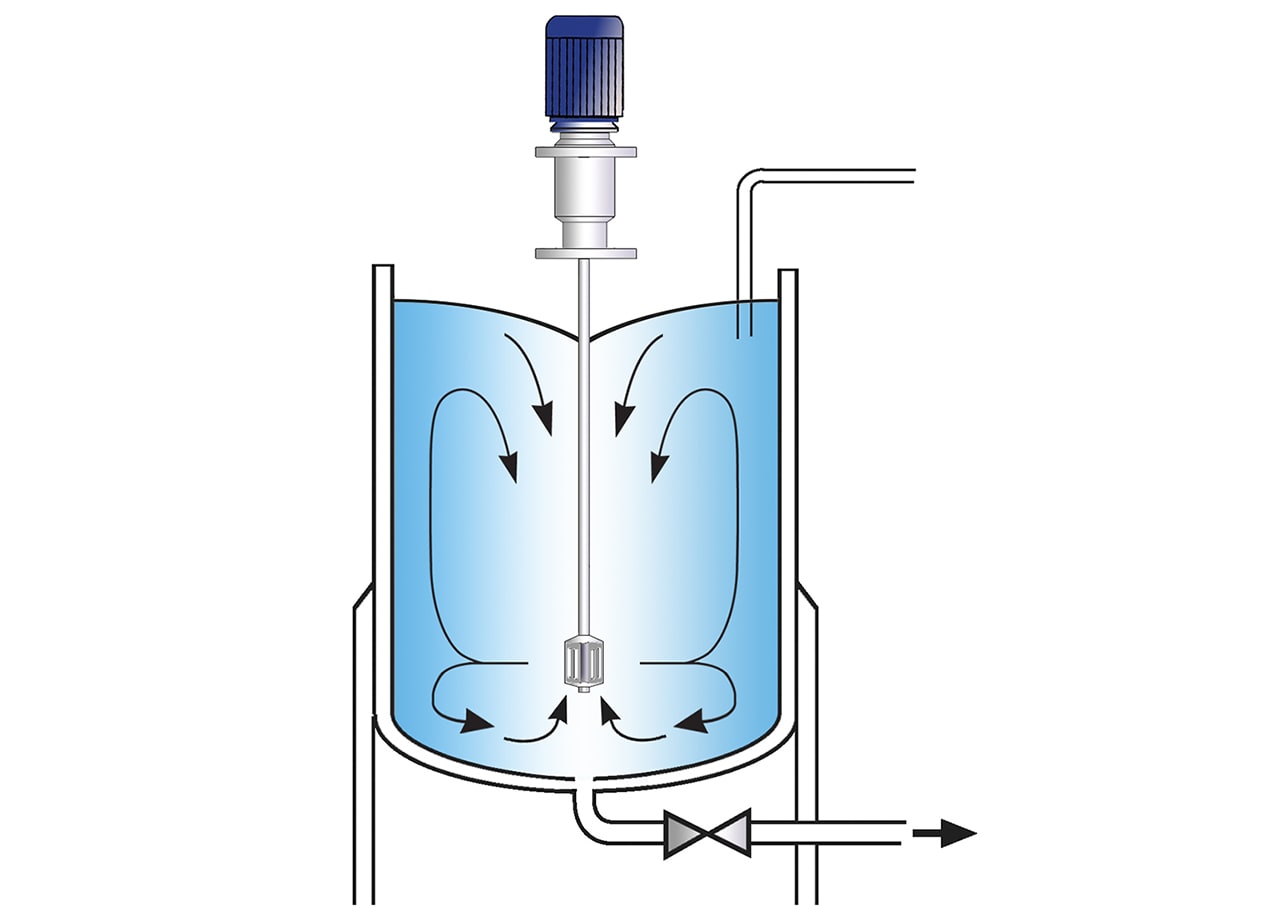
Silverson Ultramix
- Excellent in-tank movement
- Capable of rapidly incorporating large volumes of powders
- Ultra Sanitary CIP design
- Ideal for higher viscosity mixes
- Low maintenance
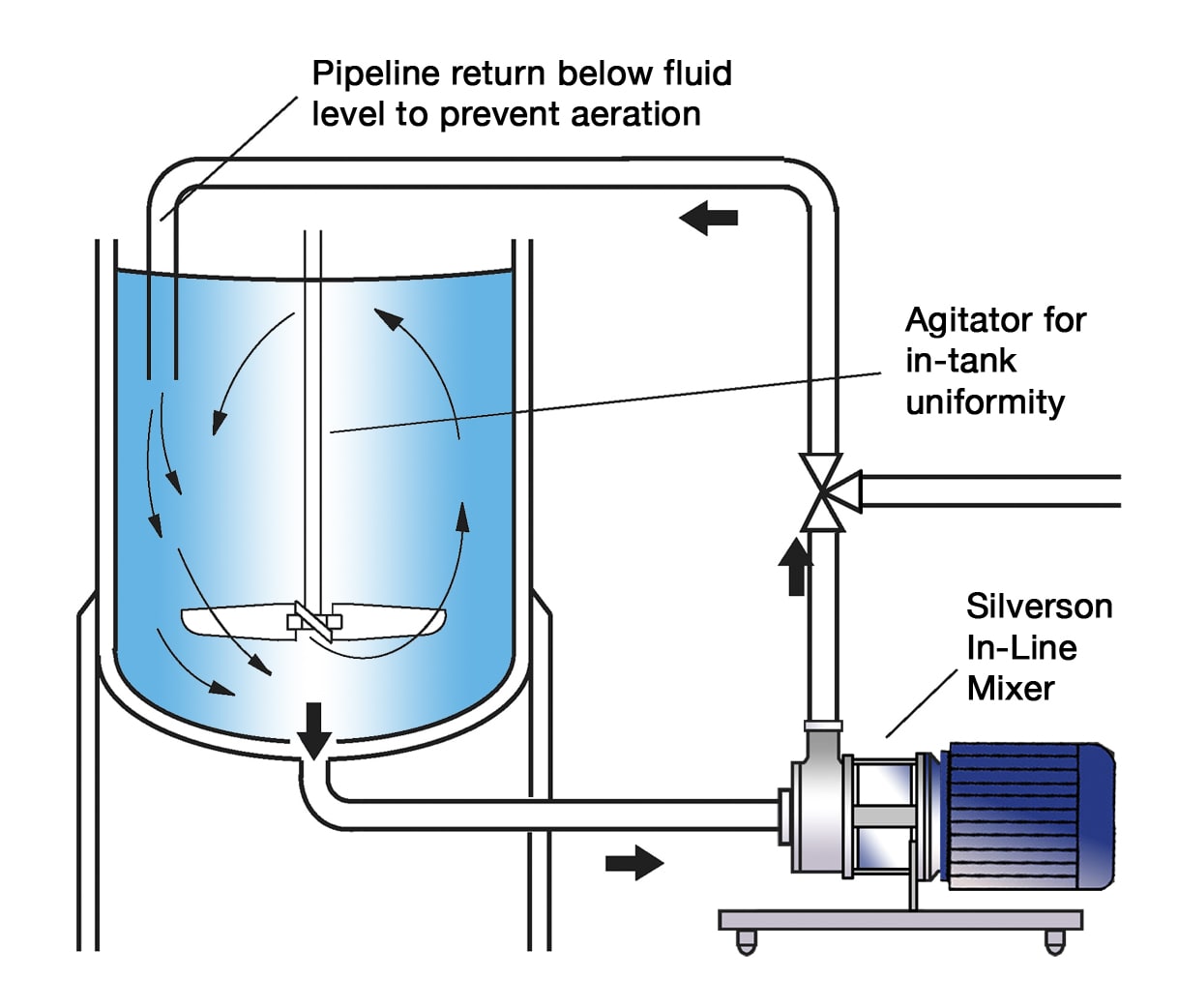
High Shear In-Line Mixers
- Ideal for larger batches
- Aeration free
- Easily retrofitted to existing plant
- Self-pumping
- Suitable for Cleaning-In-Place (CIP)
- Ultra Sanitary models available, suitable for Sterilization-In-Place (SIP)
- Can be supplied with FDA documentation
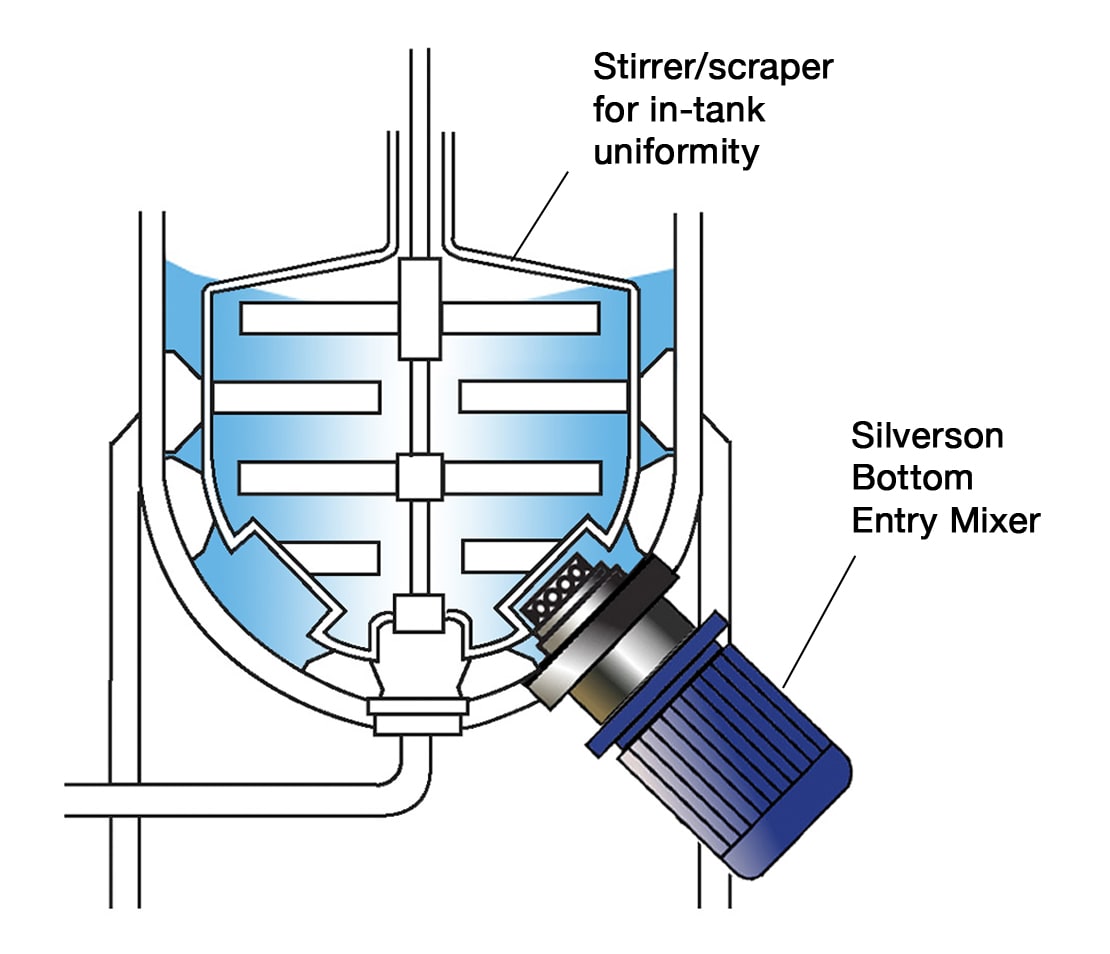
High Shear Bottom Entry Mixers
- Suitable for Cleaning-In-Place (CIP)
- Ultra Sanitary models available, suitable for Sterilization-In-Place (SIP)
- Can be supplied with FDA documentation
Silverson Flashmix
- Ideal for larger batches
- Capable of rapidly incorporating large volumes of powders
- Minimized aeration
- Suitable for Cleaning-In-Place (CIP)
- Minimum operator input required
- Easily automated
- Ultra Sanitary models available, suitable for Sterilization-In-Place (SIP)
- Can be supplied with FDA documentation